View as PDFMore than 21,000 women in the United States will receive a diagnosis of ovarian cancer (OC) in 2022. Although ovarian cancer accounts for only 1.1% of new cancer diagnoses, it accounts for 2.3% of all cancer deaths annually and has a 5-year relative survival rate of 49.1%.1 The median age at diagnosis is 63 years, with the highest incidence of ovarian cancer occurring in White women (11.3 per 100,000), followed by Asian/Pacific Islander women (9.4 per 100,000) and Black women (9.0 per 100,000).1
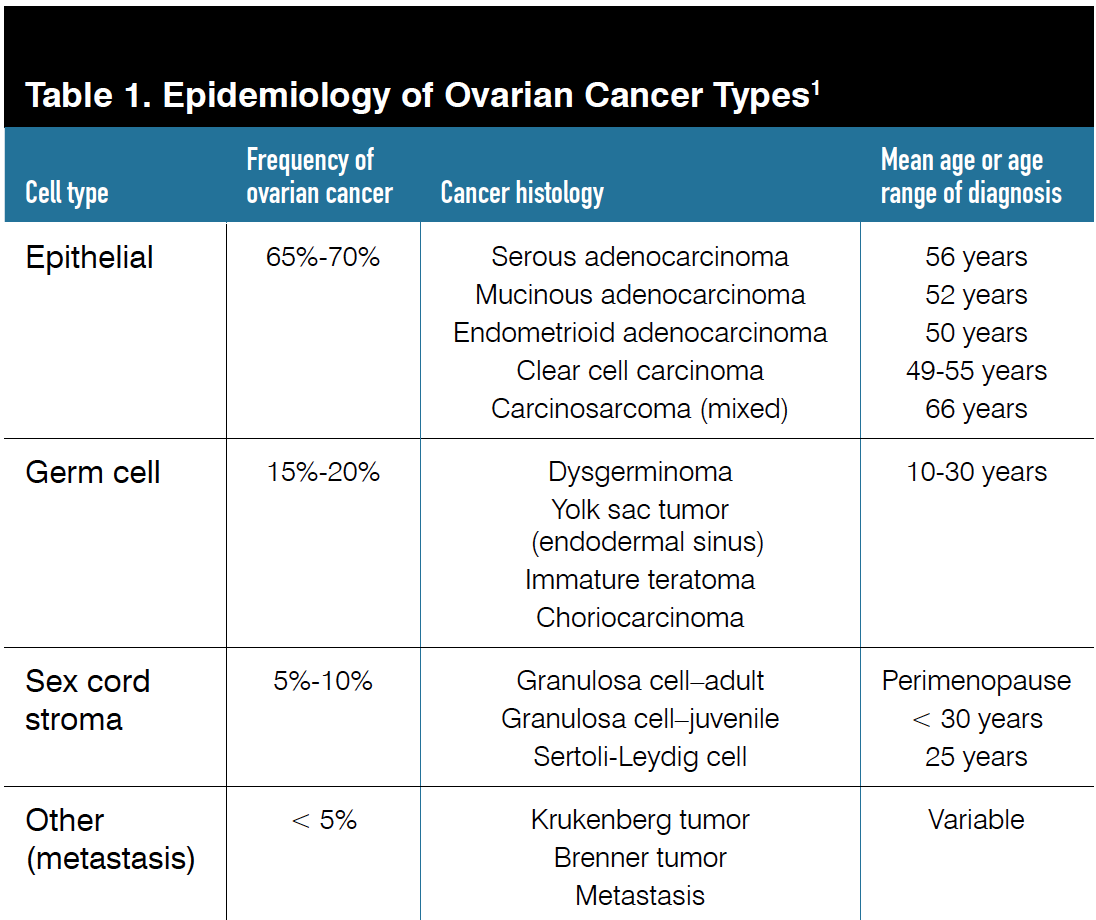
The vast majority of ovarian cancers occur in the epithelium, with a smaller fraction originating in stromal cells and germ cells (Table 1, Figure 11).
The epithelial cancers can be divided into type I and type II tumors. Whereas the type I tumors (endometrioid, clear cell, low-grade serous, and mucinous) typically behave in a more indolent fashion, the type II tumors, primarily composed of high-grade serous carcinomas, behave much more aggressively with early and widespread dissemination.2
Of all the epithelial carcinomas, high-grade serous carcinoma accounts for 60% to 80% of cases, followed by mucinous carcinoma (5%-10%), endometrioid carcinoma (7%-10%), clear cell carcinoma (8%-12%), carcinosarcoma (5%), and other more rarer histologies.3
Symptoms associated with ovarian cancer can be vague. Both the type I and II cancers are associated with abdominal pain, bloating, or gastrointestinal symptoms such as a change in bowel habits.4 Patients with symptoms should undergo pelvic/abdominal imaging and serum cancer antigen (CA) 125 testing.
Takeaways
- Theories suggest that incessant ovulation, inflammation, and tubal lesions contribute to the development of ovarian cancer.
- Approximately 18% to 24% of patients who receive a diagnosis of ovarian cancer have an inherited
germline mutation. - Risk-reducing bilateral salpingo-oophorectomy in patients with a predisposition to ovarian cancer decreases the risk of ovarian cancer by up to 95%.
- In the general population, combined oral contraceptive use for a duration of at least 5 years can reduce the incidence of ovarian cancer by up to 50%.
Unfortunately, there are no proven diagnostic screening tests (imaging or tumor markers) that have been able to improve early diagnosis or decrease disease-specific mortality.5
Origins of ovarian cancer
Although theories for development of ovarian cancer have arisen based on knowledge of risk and protective factors, understanding of the sites of origin has been enhanced by knowledge of molecular factors and genetic predisposition.
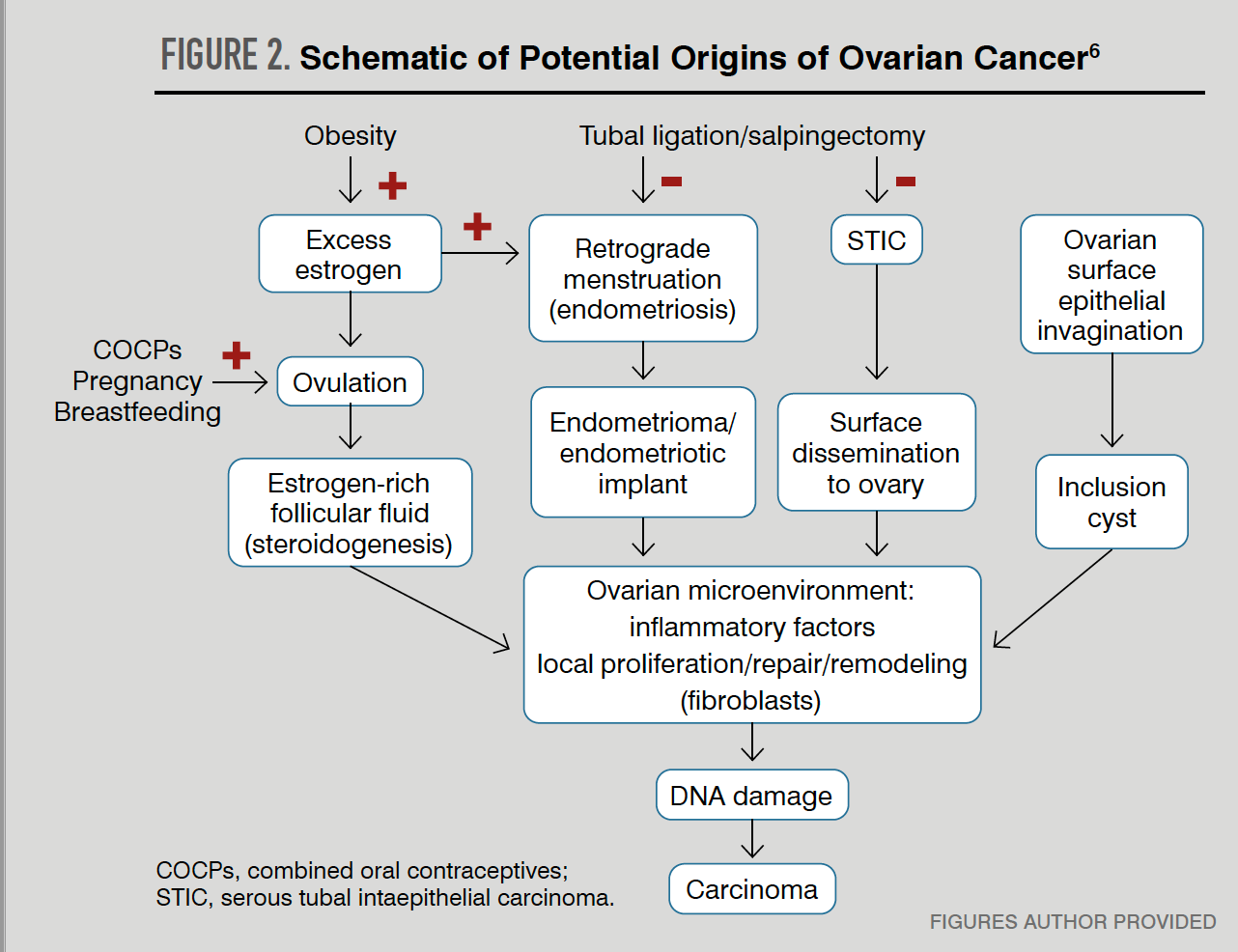
Proposed theories for ovarian carcinogenesis include incessant ovulation, gonadotropin stimulation, retrograde menstruation, and tubal inflammation. The concepts behind these theories and proposed pathways involved can overlap (Figure 2).6
Development from incessant ovulation is a hypothesis that more frequent ovulatory cycles lead to the formation and growth of dominant follicles that produce high levels of estrogen-rich fluid, induce minor trauma from the act of ovulation, and result in surrounding epithelial proliferation, inflammation, wound repair, and increased amounts of DNA damage. Cumulatively, these events are believed to contribute to premalignant transformation.
Epidemiologic studies that demonstrate reduced risk of ovarian cancer in women who experience fewer ovulatory cycles due to breastfeeding, pregnancy, or oral contraceptive use provide the most convincing evidence.7,8
The same sequence of inflammation and wound repair supports the inclusion cyst theory of ovarian cancer. This theory proposes that ovarian cancer is the result of ovarian surface epithelium invagination into stromal inclusion cysts that are surrounded by a gonadotropin-rich environment that may induce precursor lesions to become cancer.9
Lack of consistent and definitive evidence placing patients undergoing in vitro fertilization at significantly increased risk of ovarian cancer, however, does not bolster the incessant ovulation theory.10
Convincing evidence points to the fallopian tube as the origin of many ovarian cancer cases. A group of Dutch investigators first reported serous tubal intraepithelial carcinomas (STICs) lesions in prophylactically removed fallopian tubes, many taken from women with BRCA1 mutations.
The most convincing evidence of STIC as a precursor to ovarian cancer is the similar morphologic and molecular (TP53 mutation) profiles of the STICs and high grade serous ovarian carcinoma lesions.11 Up to 60% of fallopian tubes in patients with ovarian cancer demonstrate STIC at the time of hysterectomy or previous gynecologic surgery, demonstrating a role as a precursor.2
Chronic inflammation of the tubes has also been proposed as carcinogenic based on exposure of the fallopian tube epithelium to retrograde menstruation or infectious and inflammatory agents.12
Large population studies have demonstrated that occluding the tube via tubal ligation or the patient having had a previous salpingectomy can decrease the incidence of ovarian cancer by 28% and 35% (HR, 0.72, 0.65, respectively).13
Endometrioid and clear cell ovarian epithelial cancers are known to be associated with endometriosis. Because of their anatomic location, the fallopian tubes are thought to be integral to the development of endometriosis-related cancers, as endometriotic foci are believed to pass through the tubes to reach the adnexa.
Previous studies have identified occlusive tubal ligation as protective against development of endometrioid/clear cell etiologies, reporting up to a 50% reduction in these histologies, which supports the theory of endometriotic efflux as a potential contributor to nonserous ovarian histologies.14
Although endometriosis itself is not considered a premalignant condition, there are many components of endometriosis that make malignant transformation plausible. For instance, endometriotic lesions are associated with chronic inflammation and cytokine release that are thought to precipitate the malignant transformation of implants to cancer.
Because of overlapping risk factors between endometriosis and ovarian cancer (early menarche, late menopause, nulliparity), the rate of ovarian cancer among patients with known endometriosis is higher than that of the general population.
A Swedish study found that compared with controls, patients with endometriosis were 1.3 to 2.0 times more likely to develop ovarian cancer and to receive a diagnosis at a younger age.15 Fortunately, given the propensity for development of symptoms, the majority of patients who receive a diagnosis of endometriosis-associated ovarian cancer—commonly endometrioid or clear cell histologies—frequently have stage I disease as compared with the general population who receive a diagnosis of ovarian cancer at stage III or above.16
General population risk and risk factors
The overall lifetime risk of developing ovarian cancer in the general population is 1.3% to 1.5%, with a majority of cases occurring in the later years of life.1 Age and family history are the most important risk factors.
Events that decrease the number of ovulatory events are known to reduce the risk of ovarian cancer, such as the number of pregnancies (increased protection with more pregnancies), breastfeeding, and use of oral contraceptives.8 Taking combined oral contraceptives (COCPs) that avert ovulation for at least 5 years can reduce a woman’s risk of ovarian cancer by up to 50% over a period of 30 years.7
Genetic predisposition to ovarian cancer
Approximately 18% to 24% of patients with ovarian cancer have a genetic predisposition due to inheritance of germline mutations. The widespread use of germline testing on patients with incident cancers has led to the identification of an increasing number of genes associated with cancer. In addition to BRCA1 and BRCA2, BRIP, RAD51C, RAD51D, and PALB2 have been identified as genes associated with breast or ovarian cancer.
Deleterious mutations in these genes are believed to occur in only 1% of families with a history of ovarian cancer, but if these mutations are present, patients have a 2.5% to 6.0% lifetime risk of developing serous ovarian cancer.17 Although this risk is higher than that of the general population, most of this cancer risk manifests at older ages.
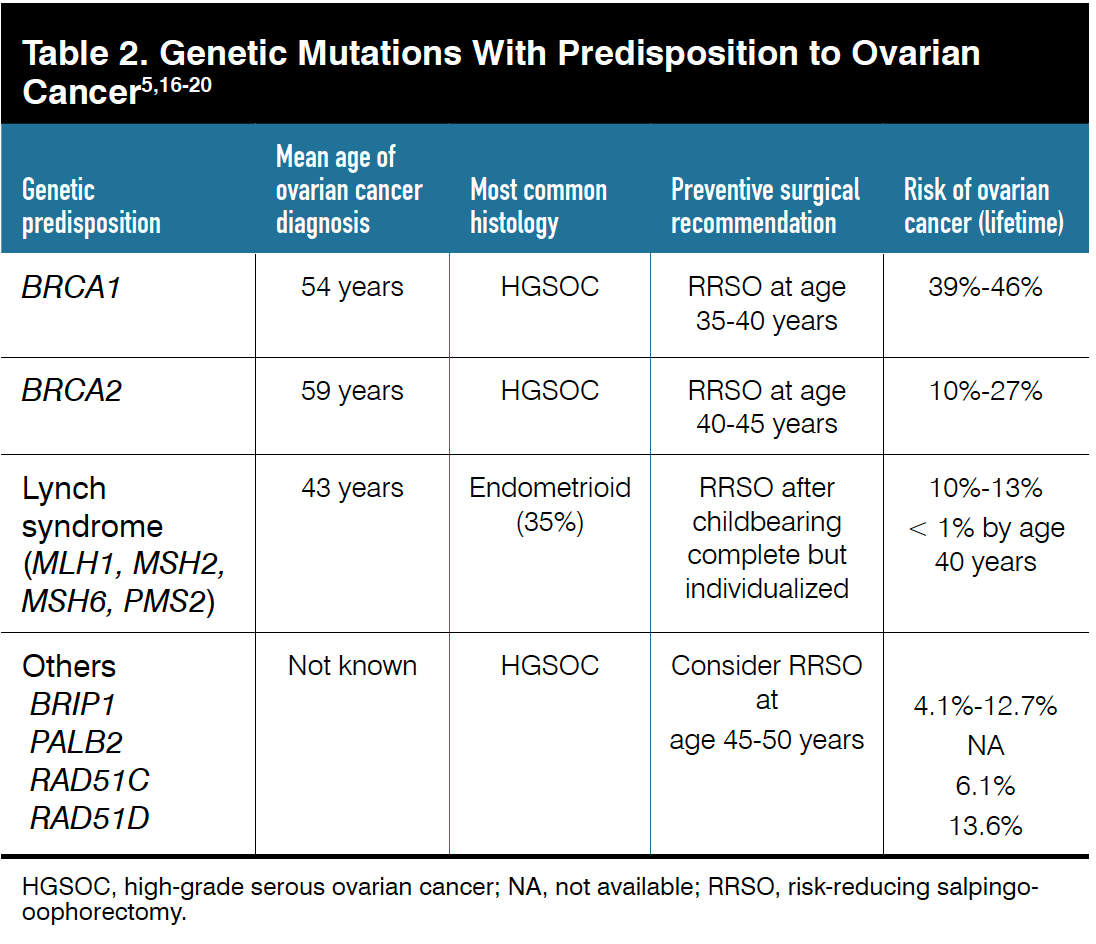
For this reason, risk-reducing salpingo-oophorectomy (RRSO) is typically recommended for patients aged 45 to 50 years.17,18 A summary of genetic mutations and overall risk for development of associated cancers is included in Table 25,16-20.
BRCA1/2
The most common germline mutations associated with the development of ovarian or fallopian tube cancer are in BRCA1 or BRCA2, genes involved in the homologous recombination DNA repair pathway. These mutations are propagated in an autosomal dominant fashion. Approximately 16% to 18% of patients with ovarian serous carcinoma have a germline BRCA mutation.19
The risk of ovarian cancer for BRCA carriers increases with age. In BRCA1 carriers, approximately 2% to 3% of cancers develop before 40 years of age, but this incidence increases to 20% by age 50, and increases even more after age 50. Hence, recommendations are for BRCA1 carriers to undergo bilateral RRSO when they are aged 35 to 40 years.19
The risk of ovarian cancer in patients with BRCA2 disease [mutation] is lower than in those with BRCA1 disease [mutation], with approximately 2% to 3% of cases occurring by age 50, so RRSO is recommended for women aged 40 to 45 years. RRSO has been found to decrease the risk of OC by 75% to 95% but also contributes to the reduction in hormone production that is protective against development of breast cancer.20 Although the absolute risk of ovarian cancer is decreased, there is still a small risk of peritoneal carcinoma in BRCA carriers.
A trial (GOG-0199) by the Gynecologic Oncology Group reported a 2.6% rate of occult STIC or small invasive carcinomas in patients at increased risk who underwent RRSO. This number is thought to reflect the residual risk of peritoneal carcinoma that was not initially detected from RRSO procedures.21
Lynch Syndrome
Patients with Lynch syndrome (hereditary nonpolyposis colorectal cancer syndrome) are at high risk of developing colorectal cancer and endometrial cancer (up to 70%) in addition to a lifetime risk of developing ovarian cancer that ranges from 10% to 13%.22
Patients with Lynch syndrome are more likely to develop lower-grade, epithelial histology ovarian cancer and are more likely to develop early-stage disease (85% are stage I or II at diagnosis). Because of their lifetime risk of developing ovarian cancer, RRSO is recommended at age 40 or after completion of childbearing, as the risk of ovarian cancer prior to age 40 is less than 1%.23
Clinical implications
Understanding the origins of ovarian/fallopian tube cancer and subsequent risk of development of cancer allows for preemptive intervention regardless of whether the patient is desirous of future fertility.
Modifiable risk factors
Although there are no guidelines on preventive interventions to reduce the risk of endometriosis-associated cancers, hormone use is recommended for endometriosis-related pelvic pain with the goal of reducing estrogen input.
Reduction in estrogen exposure to decrease ovulation and endometriotic implant stimulation can theoretically interrupt the inflammatory cascade thought to contribute to ovarian cancer precursors. In the population at increased risk for ovarian cancer, COCP use has been associated with a decreased risk of ovarian cancer in BRCA1 carriers.24 However, this use must be weighed against the possible risk of breast cancer, which is still unknown.
Genetic counseling and cascade testing
The largest impact in terms of risk reduction occurs in patients or their family members with deleterious mutations conferring increased risk. Germline testing of patients with a new diagnosis of ovarian or fallopian tube cancer can identify mutations that lead to preventive measures and cascade testing of family members. Becauase of multifactorial causes, uptake of genetic testing has been much less frequent than recommended.
Surgical risk reduction
For women with a family history or genetic predisposition, recommendations exist for prophylactic surgery.
For BRCA1 carriers, given the risk of ovarian cancer at earlier age onset, RRSO is recommended for women aged 35 to 40 years.
For BRCA2 carriers, RRSO should occur between the ages of 40 and 45 years. For other genes (BRIP, MLH1, MSH2, RAD51C, RAD51D) where there is a less than 10% risk of developing ovarian cancer, RRSO is recommended for patients aged 45 to 50 years.18 The risk of occult malignancy at the time of surgery for BRCA1 and BRCA2 carriers is approximately 2% to 10%.
For women at average risk for ovarian cancer, salpingectomy at the time of hysterectomy for other indications or for sterilization may be protective. Data in non–BRCA gene carriers who underwent tubal ligation or salpingectomy have demonstrated up to a 35% risk reduction for ovarian cancer.13
But in women 50 years or younger, the risk of all-cause mortality makes salpingo-oophorectomy less advisable unless there is a significant family history of ovarian cancer.25
This, in conjunction with the knowledge that a significant proportion of cancers may arise from the fallopian tubes, raises the question of salpingectomy for risk reduction. The prospective SOROCk trial (NCT04251052) examining elective bilateral salpingo-oophorectomy (BSO) or salpingectomy in BRCA1 carriers is underway to assess this.
Bilateral RRSO in women with a genetic predisposition or family history of ovarian cancer has been shown to decrease the risk of ovarian cancer by up to 95%.20
Approximately 3% to 8% of women who undergo RRSO for genetic predisposition will have an incidental STIC lesion found on permanent pathology, but women who have BSO for benign indications have incidental rates of less than 1%.26 If STIC lesions are identified incidentally, comprehensive evaluation of the abdominal cavity and peritoneal washings should be performed to ensure there is no evidence of disseminated disease.
However, there are no definitive recommendations for surgical staging if a STIC lesion is incidentally found; in fact, the largest study examining outcomes in isolated STIC in RRSO patients reports limited utility of surgical staging because clinical outcomes associated with observation were favorable.27 Once identified, management with surveillance pelvic ultrasound and CA125 testing are reasonable approaches to monitoring.
Conclusion
The most important aspects of gynecologic care in the prevention of ovarian/fallopian tube cancer are extensively reviewing family history and facilitating cascade genetic testing for patients and families identified with genetic mutations.
Use of COCPs is associated with risk reduction even in carriers of genetic mutation and curb inflammation that contributes to risk in disease entities. Salpingectomy has been associated with reduced risk of cancer but its contribution compared with RRSO is currently under study in carriers of genetic mutations.
References
- Cancer stat facts: ovarian cancer. National Cancer Institute Surveillance, Epidemiology, and End Results Program. Accessed November 25, 2021. https://seer.cancer.gov/statfacts/html/ovary.html
- Kurman RJ, Shih IeM. The origin and pathogenesis of epithelial ovarian cancer: a proposed unifying theory. Am J Surg Pathol. 2010;34(3):433-443. doi:10.1097/PAS.0b013e3181cf3d79. PMID:20154587; PMCID:PMC2841791
- Seidman JD, Vang R, Ronnett BM, Yemelyanova A, Cosin JA. Distribution and case-fatality ratios by cell-type for ovarian carcinomas: a 22-year series of 562 patients with uniform current histological classification. Gynecol Oncol. 2015;136(2):336-340. doi:10.1016/j.ygyno.2014.12.018. Epub 2014 Dec 17. PMID:25528497
- Dilley J, Burnell M, Gentry-Maharaj A, et al. Ovarian cancer symptoms, routes to diagnosis and survival - population cohort study in the ‘no screen’ arm of the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS). Gynecol Oncol. 2020;158(2):316-322. doi:10.1016/j.ygyno.2020.05.002. Epub 2020 Jun 17. PMID:32561125; PMCID:PMC7453382
- Menon U, Gentry-Maharaj A, Burnell M, et al. Ovarian cancer population screening and mortality after long-term follow-up in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS): a randomized controlled trial. Lancet. 2021;397(10290):2182-2193.doi:10.1016/S0140-6736(21)00731-5. Epub 2021 May 12. PMID:33991479; PMCID:PMC8192829
- Otsuka I. Mechanisms of high-grade serous carcinogenesis in the fallopian tube and ovary: current hypotheses, etiologic factors, and molecular alterations. Int J Mol Sci. 2021;22(9):4409.doi:10.3390/ijms22094409. PMID:33922503; PMCID:PMC8122889
- Collaborative Group on Epidemiological Studies of Ovarian Cancer; Beral V, Doll R, Hermon C, Peto R, Reeves G. Ovarian cancer and oral contraceptives: collaborative reanalysis of data from 45 epidemiological studies including 23,257 women with ovarian cancer and 87,303 controls. Lancet. 2008;371(9609):303-314. doi:10.1016/S0140-6736(08)60167-1. PMID:18294997
- Babic A, Sasamoto N, Rosner BA, et al. Association between breastfeeding and ovarian cancer risk. JAMA Oncol. 2020;6(6):e200421. doi:10.1001/jamaoncol.2020.0421. Epub 2020 Jun 11. PMID:32239218; PMCID:PMC7118668
- Scully RE. Pathology of ovarian cancer precursors. J Cell Biochem Suppl. 1995;23:208-218. doi:10.1002/jcb.240590928. PMID:8747398
- Rizzuto I, Behrens RF, Smith LA. Risk of ovarian cancer in women treated with ovarian stimulating drugs for infertility. Cochrane Database Syst Rev. 2019;6:CD008215. doi:10.1002/14651858.CD008215.pub2. Update in: Cochrane Database Syst Rev. 2019 Jun 18;6:CD008215. PMID:23943232; PMCID:PMC6457641
- Piek JM, van Diest PJ, Zweemer RP, et al. Dysplastic changes in prophylactically removed fallopian tubes of women predisposed to developing ovarian cancer. J Pathol. 2001;195(4):451-456. doi:10.1002/path.1000. PMID:11745677
- Salvador S, Gilks B, Köbel M, Huntsman D, Rosen B, Miller D. The fallopian tube: primary site of most pelvic high-grade serous carcinomas. Int J Gynecol Cancer. 2009;19(1):58-64.doi:10.1111/IGC.0b013e318199009c. PMID:19258943
- Falconer H, Yin L, Grönberg H, Altman D. Ovarian cancer risk after salpingectomy: a nationwide population-based study. J Natl Cancer Inst. 2015;107(2):dju410. doi:10.1093/jnci/dju410. PMID:25628372
- Sieh W, Salvador S, McGuire V, et al; Ovarian Cancer Association Consortium. Tubal ligation and risk of ovarian cancer subtypes: a pooled analysis of case-control studies. Int J Epidemiol. 2013;42(2):579-589.doi:10.1093/ije/dyt042. PMID:23569193; PMCID:PMC3619957
- Melin A, Sparén P, Persson I, Bergqvist A. Endometriosis and the risk of cancer with special emphasis on ovarian cancer. Hum Reprod. 2006;21(5):1237-1242.doi:10.1093/humrep/dei462. Epub 2006 Jan 23. PMID:16431901
- Sainz de la Cuesta R, Eichhorn JH, Rice LW, Fuller AF Jr, Nikrui N, Goff BA. Histologic transformation of benign endometriosis to early epithelial ovarian cancer. Gynecol Oncol. 1996;60(2):238-244.doi:10.1006/gyno.1996.0032. PMID:8631545
- Tung N, Domchek SM, Stadler Z, et al. Counselling framework for moderate-penetrance cancer-susceptibility mutations. Nat Rev Clin Oncol. 2016;13(9):581-588. doi:10.1038/nrclinonc.2016.90. Epub 2016 Jun 14. PMID:27296296; PMCID:PMC5513673
- Song H, Dicks E, Ramus SJ, et al. Contribution of germline mutations in the RAD51B, RAD51C, and RAD51D genes to ovarian cancer in the population. J Clin Oncol. 2015;33(26):2901-2907.doi:10.1200/JCO.2015.61.2408. Epub 2015 Aug 10. PMID:26261251; PMCID:PMC4554751
- Zhang S, Royer R, Li S, et al. Frequencies of BRCA1 and BRCA2 mutations among 1,342 unselected patients with invasive ovarian cancer. Gynecol Oncol. 2011;121(2):353-357. doi:10.1016/j.ygyno.2011.01.020. Epub 2011 Feb 15. PMID:21324516
- Kauff ND, Satagopan JM, Robson ME, et al. Risk-reducing salpingo-oophorectomy in women with a BRCA1 or BRCA2 mutation. N Engl J Med. 2002;346(21):1609-1615. doi:10.1056/NEJMoa020119. Epub 2002 May 20. PMID:12023992
- Sherman ME, Piedmonte M, Mai PL, et al. Pathologic findings at risk-reducing salpingo-oophorectomy: primary results from Gynecologic Oncology Group Trial GOG-0199. J Clin Oncol. 2014;32(29):3275-3283. doi:10.1200/JCO.2013.54.1987. Epub 2014 Sep 8. PMID:25199754; PMCID:PMC4178524
- Watson P, Bützow R, Lynch HT, et al. The clinical features of ovarian cancer in hereditary nonpolyposis colorectal cancer. Gynecol Oncol. 2001;82(2):223-228.doi:10.1006/gyno.2001.6279. PMID:11531271
- Bonadona V, Bonaïti B, Olschwang S, et al. Cancer risks associated with germline mutations in MLH1, MSH2, and MSH6 genes in Lynch syndrome. JAMA. 2011;305(22):2304-2310.doi:10.1001/jama.2011.743. PMID:21642682
- Schrijver LH, Antoniou AC, Olsson H, et al; Hereditary Breast and Ovarian Cancer Research Group Netherlands and International BRCA1/2 Carrier Cohort Study. Oral contraceptive use and ovarian cancer risk for BRCA1/2 mutation carriers: an international cohort study. Am J Obstet Gynecol. 2021;225(1):51.e51-51.e17.doi:10.1016/j.ajog.2021.01.014. Epub 2021 Jan 22. PMID:33493488; PMCID:PMC8278569
- Cusimano MC, Ferguson SE, Moineddin R, et al. Ovarian cancer incidence and death in average-risk women undergoing bilateral salpingo-oophorectomy at benign hysterectomy. Am J Obstet Gynecol. 2022;226(2):220.e1-220.e26. doi:10.1016/j.ajog.2021.09.020doi:10.1016/j.ajog.2021.09.020. Epub 2021 Sep 24. PMID:34563499
- Rabban JT, Garg K, Crawford B, Chen LM, Zaloudek CJ. Early detection of high-grade tubal serous carcinoma in women at low risk for hereditary breast and ovarian cancer syndrome by systematic examination of fallopian tubes incidentally removed during benign surgery. Am J Surg Pathol. 2014;38(6):729-742. doi:10.1097/PAS.0000000000000199. PMID:24820399
- Wethington SL, Park KJ, Soslow RA, et al. Clinical outcome of isolated serous tubal intraepithelial carcinomas (STIC). Int J Gynecol Cancer. 2013;23(9):1603-1611.doi:10.1097/IGC.0b013e3182a80ac8. PMID:24172097; PMCID:PMC4979072