Woman With Heterotopic Pregnancy After Natural Conception
A 29-year-old gravida 10, para 3 (1 term gestation, 1 preterm gestation of twins, 1 stillbirth at 5 months, 2 spontaneous abortions, and 4 elective abortions) presented to the clinic at about 5 weeks’ gestation with abdominal pain and vaginal bleeding. She described the pain as sporadic, mostly on the left side, exacerbated by movement, and resolving with rest, and the bleeding as initially intermittent but then heavier “like a period.”
A 29-year-old gravida 10, para 3 (1 term gestation, 1 preterm gestation of twins, 1 stillbirth at 5 months, 2 spontaneous abortions, and 4 elective abortions) presented to the clinic at about 5 weeks’ gestation with abdominal pain and vaginal bleeding. She described the pain as sporadic, mostly on the left side, exacerbated by movement, and resolving with rest, and the bleeding as initially intermittent but then heavier “like a period.” Abdominal and bimanual findings were normal. Speculum examination revealed scant blood in the vaginal vault, the os was closed; there was no visible tissue. Hematocrit level was 39%. A quantitative human chorionic gonadotropin (hCG) test was ordered but not obtained, for unknown reasons. Same-day ultrasonogram showed a single intrauterine gestational sac with a yolk sac but no visualized embryo; the adnexae were unremarkable. After a period of observation, she was released with instructions to go to the emergency department (ED) if the bleeding increased or the pain worsened. She returned to the clinic 3 days later because of vaginal spotting. Abdominal findings remained otherwise unremarkable, and she was given the same instructions.
At 6 weeks’ gestation, she awoke with nausea and severe abdominal pain and went immediately to the ED. She had light vaginal bleeding and new onset anorexia. She denied fever, chills, and dysuria. On examination, she was in severe distress and appeared moderately ill. Temperature was 35.9°C (96.6°F); blood pressure, 134/74 mm Hg; heart rate, 70 beats per minute; and respiration rate, 28 breaths per minute. Abdominal palpation elicited right lower quadrant tenderness, with rebound and guarding.
Ultrasonographic imaging revealed a viable intrauterine pregnancy (IUP) and an ectopic pregnancy (EP) in the right adnexa (Figure 1). Cardiac activity was noted within the adnexal mass.
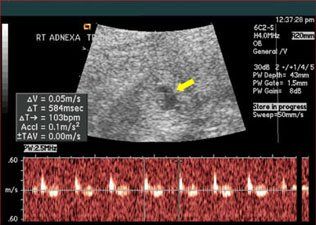
Figure 1 – This ultrasonogram shows a viable intrauterine pregnancy and an ectopic pregnancy in the right adnexa. Cardiac activity was noted within the adnexal mass. Taken at 6 weeks.
HETEROTOPIC PREGNANCY: AN OVERVIEW
Heterotopic pregnancy (HP) is the existence of 2 (or more) simultaneous pregnancies with separate implantation sites, one of which is ectopic.
Incidence. HP is estimated to occur in 1 in 10,000 to 30,000 pregnancies.1 In contrast, EP occurs in about 2% of all pregnancies in the United States.2 The incidence of HP appears to be higher in certain subsets, in particular women who have been treated for infertility with some form of artificial reproductive technology (ART).3 Patients who have been treated with ART have about a 1% risk of HP and typically undergo routine early ultrasonographic screening for this condition.3 HP is otherwise rare in women with spontaneous gestation, such as this patient, and routine early ultrasonographic screening is not recommended for this population.
Risk factors. In a review of 80 cases of HP from 1994 to 2004, 35% of patients had a history of a EP, 33.75% had salpingectomy, 31.25% had a history of pelvic inflammatory disease (PID), and 23.75% had reconstructive tubal surgery.4 Most of the cases were also related to ART treatment; however, 13 of the 80 patients had an HP after a spontaneous pregnancy, and of these, 11 had no underlying risk factors.4 This patient had no history of ART treatment; however, she did have a history of tobacco use (1 pack per day), which is a risk factor for EP.5 She had no other risk factors for EP (ie, a history of EP, tubal surgery, in utero diethylstilbestrol exposure, PID, or recurrent chlamydia or gonorrhea infections).5,6
Location. Similar to EP, most cases of HP occur in the fallopian tube; other commonly affected sites include the cervix and a cesarean delivery scar.4 HP occurring in the tube stump and cornual region has also been reported.3
Morbidity and mortality. EP remains the leading cause of maternal death in the first trimester1 and accounts for 10% to 15% of all maternal deaths.7 However, the risk of later presentation and life-threatening hemorrhage and hemorrhagic shock is high in HP because of its diagnostic challenges.3 Most cases of HP are diagnosed after rupture of the EP component.4 Tubal rupture and hemorrhage are most common for HP located in the ampulla.4 Despite the associated morbidity, more than half of cases of HP have resulted in successful term deliveries.4
DISTINGUISHING FEATURES AND DIAGNOSIS OF HP
Clinical manifestations. In a review of 66 cases of combined intrauterine and extrauterine gestations, abdominal pain alone was found to be the most common presenting symptom of HP.8 The combination of lower abdominal pain, an adnexal mass, peritoneal irritation, and an enlarged uterus most strongly supported the existence of HP. Vaginal bleeding was present in 21 cases.
Differential diagnosis. The main concern in this patient, with her continued vaginal spotting and known IUP, was threatened abortion. Had a pelvic examination been performed on her second visit because of persistent spotting, an adnexal mass may have been identified and HP may have been diagnosed earlier. Her clinical presentation in the ED-the abdominal pain and peritoneal signs-was consistent with HP. Abdominal pain in early pregnancy can represent a number of conditions, including:
- Ruptured EP, which classically presents as abdominal pain and vaginal bleeding after 7 weeks of amenorrhea.7 The pain usually lateralizes to one side.
- Ovarian torsion and ruptured corpus luteum cyst can both present with sudden onset lateralizing pain but usually no vaginal bleeding.
- Subchorionic hemorrhage, caused by a partial detachment of the trophoblast from the uterus, can present with pain and bleeding in early pregnancy.9 A small subchorionic hemorrhage is generally inconsequential for miscarriage risk; however, a large subchorionic hemorrhage may indicate pending miscarriage.9
- Acute appendicitis typically presents with fever, anorexia, and abdominal pain in the periumbilical region. The pain typically progresses to the right lower quadrant, without vaginal bleeding.
- Spontaneous abortion can present with pelvic pain and vaginal bleeding in early pregnancy. Typically, the pain is mid pelvic rather than localized to one side.
Ultrasonographic findings. Given the overlap in clinical presentations, imaging is critical for diagnosis. However, the findings can be misleading in HP. Clear and simultaneous visualization of both intrauterine and extrauterine pregnancies is not always possible, especially early in gestation. In patients treated with ART, ovarian hyperstimulation syndrome is a frequent occurrence that can mask the presence of an EP.4 It is not uncommon for HP to be initially misinterpreted as a luteal cyst.10 Transvaginal ultrasonogram can detect an IUP as early as 4.5 to 5 weeks’ gestation (Figure 2, was taken at around 5.5 weeks).15 Cardiac activity can be detected at 5.5 to 6 weeks.1
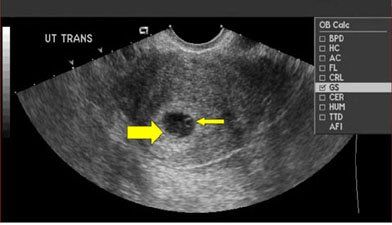
Figure 2A
Figure 2 – In a normal pregnancy, the gestational sac (A, large arrow) and yolk sac (A, small arrow) appear as a hypoechoic structure encircled by an echogenic border within the endometrial cavity at about 5.5 weeks’ gestation. This is followed by development of the embryo, alongside the yolk sac at about 5.5 to 6 weeks’ gestation (B).
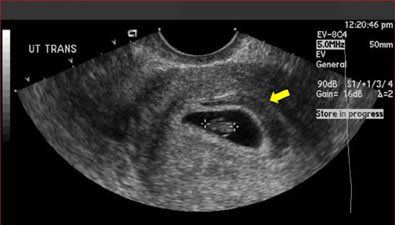
Figure 2B
Gestational age at diagnosis. The reported gestational age at diagnosis of HP ranges from 5 to 20 weeks, with a mean of 8 weeks, give or take 3 weeks.4 In a study comparing HP and tubal EP, the gestational age at diagnosis was 7.5 weeks for HP, and 7.2 weeks for EP; sonographic examination led to diagnosis in all 12 cases of HP and in 94.3% of 210 cases of EP.3 However, in a review of 80 patients with HP, ultrasonographic findings were definitively diagnostic in only 25% of patients; laparoscopy or laparotomy was diagnostic in the remaining 75% of patients.4
Quantitative hCG test results. The classic definition of a normal pregnancy, the doubling of hCG every 48 hours, has been redefined as an at least 53% increase in hCG over 48 hours.11 An abnormally slow rise in hCG levels would certainly suggest the presence of an EP. However, an EP can also have a normal hCG rise.12 In a normal spontaneous gestation, once an IUP has been identified, hCG levels are typically unnecessary.
TREATMENT AND OUTCOME
Laparoscopy is preferred over laparotomy for the treatment of HP because of its favorable profile for less bleeding, pain, hospitalization, recovery time, and cost.1 For an extrauterine gestation in a tubal location, salpingectomy is indicated over salpingostomy.1 The latter may result in incomplete removal and persistent EP, which would be impossible to identify with serial hCG levels given the existing IUP.
This patient underwent emergent laparoscopy; however, because of a large hemoperitoneum, open laparotomy was necessary. This revealed a rupture in the middle third of the right fallopian tube; the entire tube was edematous and ecchymotic. A right salpingectomy was performed. Postoperative obstetrical ultrasonogram showed a persistent viable IUP with normal fetal heart tones. The patient remained stable and was discharged 2 days after surgery. She did not return for follow-up as instructed. When contacted, she stated that she had terminated the remaining pregnancy.
Although the likelihood of a coexisting extrauterine gestation is extremely small after ultrasonographic confirmation of an IUP, this case highlights the need for a high index of suspicion for HP in patients in the first trimester, or even several weeks beyond, who present with signs and symptoms potentially consistent with an EP.
References:
REFERENCES:
1. Mukul LV, Teal SB. Current management of ectopic pregnancy. Obstet Gynecol Clin N Am. 2007; 34:403-419.
2. Eyvazzadeh AD, Levine D. Imaging of pelvic pain in the first trimester of pregnancy. Radiol Clin N Am. 2006;44:863-877.
3. Yeh HC, Goodman JD, Carr L, et al. Intradecidual sign: a US criterion of early intrauterine pregnancy. Radiology. 1986;161:463-467.
4. Barrenetxea G, Barinaga-Rementeria L, Lopez de Larruzea A, et al. Heterotopic pregnancy: two cases and a comparative review. Fertil Steril. 2007;87:417.e9-e15.
5. Ankum WM, Mol BW, Van der Veen F, et al. Risk factors for ectopic pregnancy: a meta-analysis. Fertil Steril. 1996;65:1093-1099.
6. Hillis SD, Owens LM, Marchbanks PA, et al. Recurrent chlamydial infections increase the risks of hospitalization for ectopic pregnancy and pelvic inflammatory disease. Am J Obstet Gynecol. 1997;176:103-107.
7. Lozeau AM, Potter B. Diagnosis and management of ectopic pregnancy. Am Fam Physician. 2005;72;1707-1714, 1719-1720.
8. Reece EA, Petrie RH, Sirmans MF, et al. Combined intrauterine and extrauterine gestations: a review. Am J Obstet Gynecol. 1983;146:323-330.
9. Eyvazzadeh AD, Levine D. Imaging of pelvic pain in the first trimester of pregnancy. Radiol Clin N Am. 2006; 44:863-877.
10. Garcia Oliveira F, Abdelmassih V, Eigenheer AL, et al. Rare association of ovarian implantation site for patients with heterotopic and with primary ectopic pregnancies after ICSI and blastocyst transfer. Hum Reprod. 2001;16:2227-2229.
11. Barnhart KT, Sammel MD, Rinaudo PF, et al. Symptomatic patients with an early viable intrauterine pregnancy: hCG curves redefined. Obstet Gynecol. 2004;104:50-55.
12. Silva C, Sammel MD, Zhou L, et al. Human chorionic gonadotropin profile for women with ectopic pregnancy. Obstet Gynecol. 2006;107:605-610.
Dr Pippitt is a visiting instructor and Dr Stoesser is assistant professor both in the department of family and preventive medicine at University of Utah School of Medicine.
Acknowledgment: With special thanks to Dr Michael Draper, MFM, for help with ultrasonogram interpretation and Dr Richard Chapa, Ob/Gyn for operative management and clinical consultation.
This case is to be published in an upcoming issue of Consultant.
Reducing multifetal pregnancy through publicly funded IVF programs
April 26th 2024Learn how a mandatory elective single-embryo transfer policy in publicly funded in vitro fertilization programs significantly decreases multifetal pregnancy rates, offering insights into mitigating risks in assisted reproduction.
Read More
In this episode of Pap Talk, Gloria Bachmann, MD, MSc, breaks down what it means to be a health care provider for incarcerated individuals, and explores the specific challenges women and their providers face during and after incarceration. Joined by sexual health expert Michael Krychman, MD, Bachmann also discusses trauma-informed care and how providers can get informed.
Listen
SART data indicates rise in IVF use in 2022
April 24th 2024Dive into the latest statistics from the Society for Assisted Reproductive Technology, revealing a 6% increase in in vitro fertilization cycles and a surge in egg freezing, amidst a landscape of rising fertility service demand and evolving clinical practices.
Read More
Excessive gonadotropins in IVF: Effects on mosaicism and live birth
April 12th 2024A recent study revealed a correlation between high doses and prolonged duration of exogenous gonadotropin use during in vitro fertilization and increased embryonic mosaicism alongside diminished live birth rates, prompting reconsideration of dosage and duration protocols.
Read More
